
The Cannabis Contamination Conundrum
Opiate related deaths have decreased by twenty-five percent in states with operational medicinal cannabis programs. No one has ever died as a direct result from cannabis use but that doesn’t mean that all cannabis is always 100% safe. Cannabis products span several markets, including agricultural/environmental, food safety (especially edibles and beverages) and medicine. Without proper quality control testing, dangerous pesticides, heavy metals, residual solvents, microorganisms, aflatoxins, etc. could be found in a “lab tested” medicinal cannabis product. Without nationally accepted regulations and standards this problem is further exacerbated. The solution is as simple as state commissions saying, use this analytical instrument and use it this way to get results.
Currently some states do not require quality control testing at all. The ones that do are only required to test for things like potency and contaminants, but there are no methods and standards for this testing. Growers and dispensaries may select the laboratory that consistently gives them the highest THC reported values, as products with greater THC levels are considered premium products. Consumers and patients may be paying a premium for high THC products, when in reality these cannabis products may contain much less THC than reported by the lab. Also, when growers breed high THC level cannabis, they are likely reducing the levels of other potentially beneficial cannabinoids.
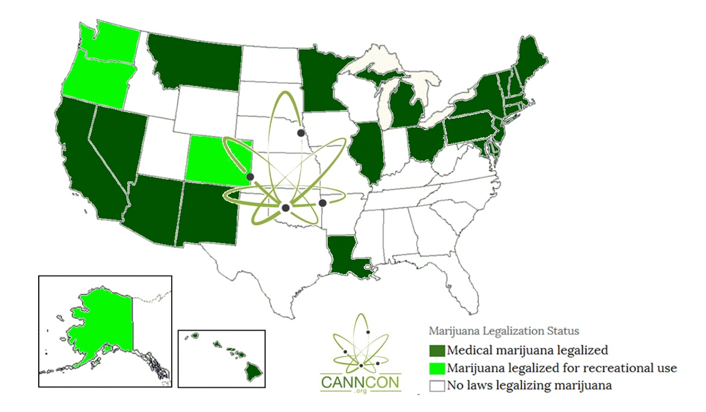
Current cannabis legalization map of the United States. Cannabis quality control testing and contamination testing requirements vary from state to state.
Low levels of cannabis contaminants may not kill a medical cannabis user, but they can significantly reduce the medical benefits that these patients would have received. Over long periods of time, even very low level exposure to toxic heavy metals, dangerous pesticides and residual solvents can cause serious health problems. A big problem here is that some of the patients using medical cannabis may be severely immunocompromised to begin with, and may become even more immunocompromised from western treatments like chemotherapy and radiation. Even infants are using cannabis as medicine now. If a dangerous pesticide or mold found its way into a very sick child’s body, it could cause extreme illness or death. The last thing that we want as a community is to make people sicker. Cannabis offers an opportunity to heal the world, but we must ensure that medical cannabis products are free from toxic contaminants.
There are already great analytical instrument/reagent manufacturers, like Shimadzu, RESTEK, Fritsch, PerkinElmer and MilliporeSigma, as well as chemical organizations, like AOCS, that have been working with quality control methods and standards in other industries like, food safety, clinical laboratories, agricultural labs and even forensics labs. It can be as simple as the state leaders looking to these great resources for guidance on medicinal cannabis methods and standards. I always use the analogy of bubble gum and aspirin. You can walk into a corner store and purchase both, and they are both tested for quality control, but aspirin is tested using more stringent methods and standards, because it is medicine. It is a problem when recreational cannabis and medicinal cannabis are being testing the same way.

Modern instruments like triple quadrupole mass spectrometers are used to determine pesticide contamination levels.
It is very important for medicinal cannabis to have higher testing standards, as it is being used as medicine to treat very sick people. So many people that are sick or have sick children that live in states without medicinal cannabis programs relocate to legal cannabis states like Colorado or Oregon, thinking that relocation is the end of their problems. Sadly, they may be unaware of cannabis contamination issues. With pesticide recalls happening every other week, obviously change is needed. Products CANNOT make their way to dispensary shelves and later be pulled for re-testing to find they are contaminated after many patients have already purchased and used these contaminated products.
This industry has a chance to self-regulate, before the government regulations come. I can tell you one thing, if we wait for the government, someone is not going to be happy with the results and it won’t be the government walking away unhappy. On top of that, most of the labs will need to purchase new instruments to meet these standards. Both Shimadzu and AOCS have immersed themselves into the cannabis market in order to apply their expertise to help with these problems. The hard part is getting labs to go above and beyond what they are currently required to do by the state they are in.
New approaches in personalized (or precision) cannabinomics offer opportunities to improve medical cannabis safety and efficacy.
I see an additional issue ahead that needs to be addressed in order to improve patient safety. If you look closely at modern cannabis cultivation and oil extraction processes, great care is taken to ensure that high-quality, contaminant-free oils are produced. These oils may be tested for potency, residual solvents, heavy metals, pesticides, aflatoxins/mycotoxins and microorganisms. While these oils are certified contaminant free, they may be then placed in untested dispensing materials, such as electronic vaporizer pens manufactured overseas. These vape pens may contain additional toxic contaminants, such as plasticizers or heavy metals. Yet nobody is testing these cannabis products in their final form.
 Also, it is important to note that when you put something through an extraction process that you are not just extracting the cannabinoids to their purest forms, but also any contaminants that may have come in contact with the sample. This is most alarming, because most pediatric cannabis patients are using extracted tinctures and oil as medicine. A lot is at stake when you are dealing with small children that could be immunocompromised. Concentrated contaminants pose extreme risk to pediatric or any immunocompromised patients. These contaminants may come from places other than the cannabis, such as the solvents used for the extraction process.
Also, it is important to note that when you put something through an extraction process that you are not just extracting the cannabinoids to their purest forms, but also any contaminants that may have come in contact with the sample. This is most alarming, because most pediatric cannabis patients are using extracted tinctures and oil as medicine. A lot is at stake when you are dealing with small children that could be immunocompromised. Concentrated contaminants pose extreme risk to pediatric or any immunocompromised patients. These contaminants may come from places other than the cannabis, such as the solvents used for the extraction process.
Our non-profit organization, CANNCON, Inc., was formed to unite the analytical science community with the medicinal cannabis community. We are devoted to the advancement of cannabis science, analytical testing for quality control, and medicinal cannabis. Personalized treatments are the future of medicinal cannabis. Potency testing, contamination testing, and strain mapping alone is not enough. We should also be looking at patient’s genomes and metabolomes to see which cannabis strains work best for different disorders/diseases based on their DNA and metabolites. A medical patient may be a fast metabolizer of a particular cannabinoid, and therefore may require higher dosing or an alternate cannabinoid for therapeutic benefit. We have started connecting sick children with experts, like Dr. Kevin Rosenblatt (CompanionDx Lab), who is a key opinion leader in genomics, metabolomics and proteomics. This personalized (or precision) approach to cannabis treatments is a new frontier in cannabis medicine.
As we move forward with cannabis based therapies and therapeutic drug monitoring approaches, it is important that we work together across a multitude of disciplines to improve cannabis quality and patient outcomes. It is unfathomable that most cannabis users know more about the health benefits of cannabis and the endocannabinoid system than most medical doctors do. Cannabis experts, medical doctors and analytical scientists must come together to share information and develop best practices. Our annual Cannabis Science Conference will be held in Portland, Oregon, October 4th through 6th, and is ideal for experts and novices alike. Together we can explore and improve cannabis science and dispel many of the myths and misconceptions regarding cannabis and further substantiate the health benefits on medicinal cannabis.
Joshua Crossney is the President & Founder of CANNCON, Inc., a 501(c)3 non-profit organization devoted to the advancement of cannabis science, analytical testing, and personalized (percision) medicine. Joshua has a backgound in analytical science recruiting …
